
MEL Chemistry Lite is a subscription service from MEL Science offering monthly deliveries of safe chemistry experiments for kids. Each box is $19.90 per month + free shipping, and will include one hands-on experiment + FREE VR lessons you can access via the MEL VR app! Mel Lite is currently available in 5 countries: USA, UK, Chile, Indonesia, and India.

The box sleeve indicates the experiment for this month and a list of the materials.

The box welcomes us to science in different languages!

We’re so excited to finally open this box!

The inner lid has an instruction on how to put a hole in the bottle’s nozzle.
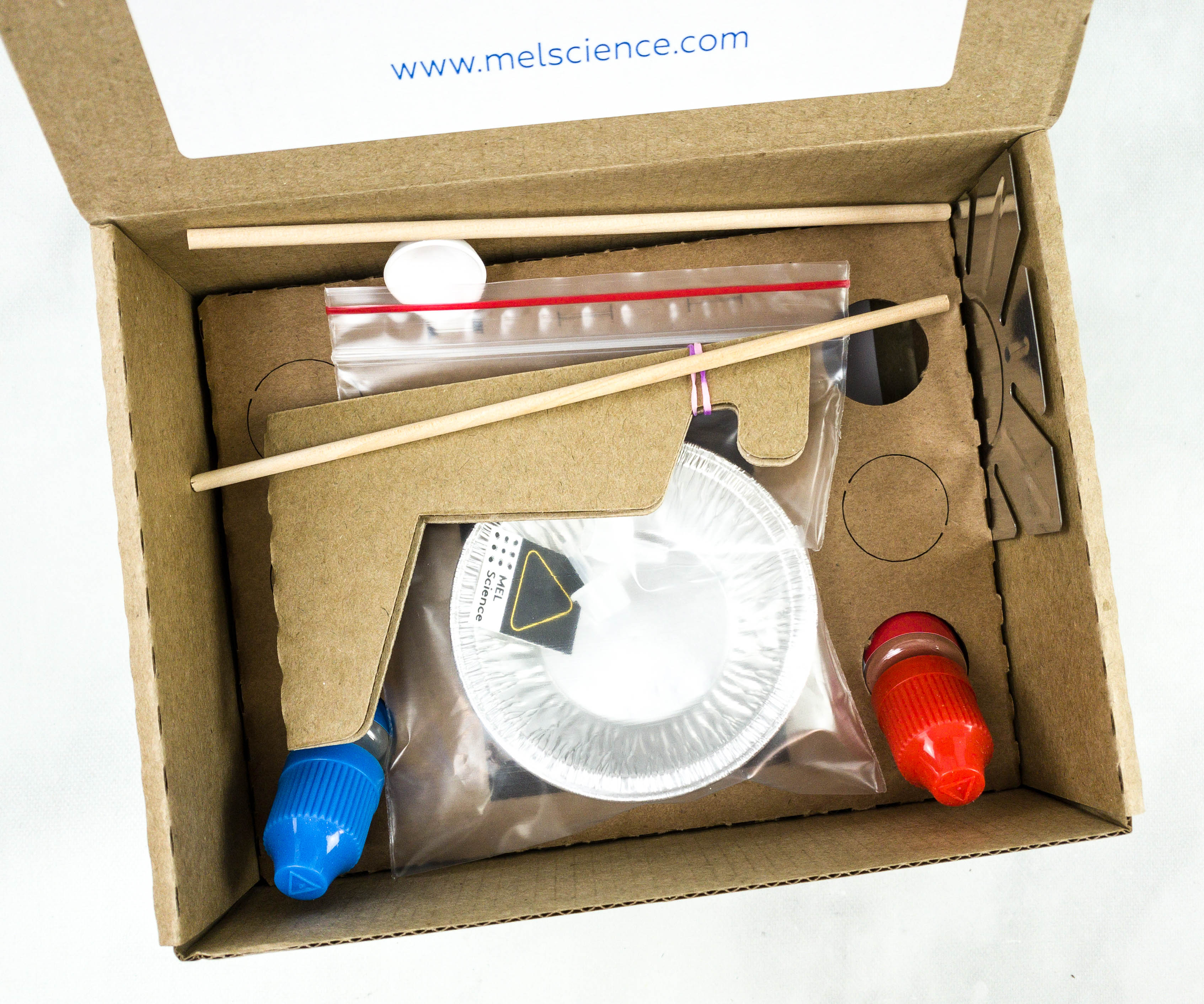
The items were packed with a carton to keep them in place during shipping.

A QR code is also available at the back of the card for easy download.

Everything in my box!

The box included this experiment card. It shows us the level of difficulty and danger, as well as the duration.

At the back of the card, there’s an explanation on how the experiment works!
When things burn, and metals are no exception, more often than not some rogue particles – ions and atoms – leave the burning material for the air. When they first appear, they’re in a so-called “excited state,” meaning that some of their electrons are quick to settle more “conveniently” though, and as they do so, they emit some light.
One interesting thing about the light is that its color is very specific to the kind of atom or ion that emitted it. That’s why different metals tint the flame of different colors. Zinc makes blue, copper makes green, etc. You can probably guess how they make fireworks in all sorts of different colors!
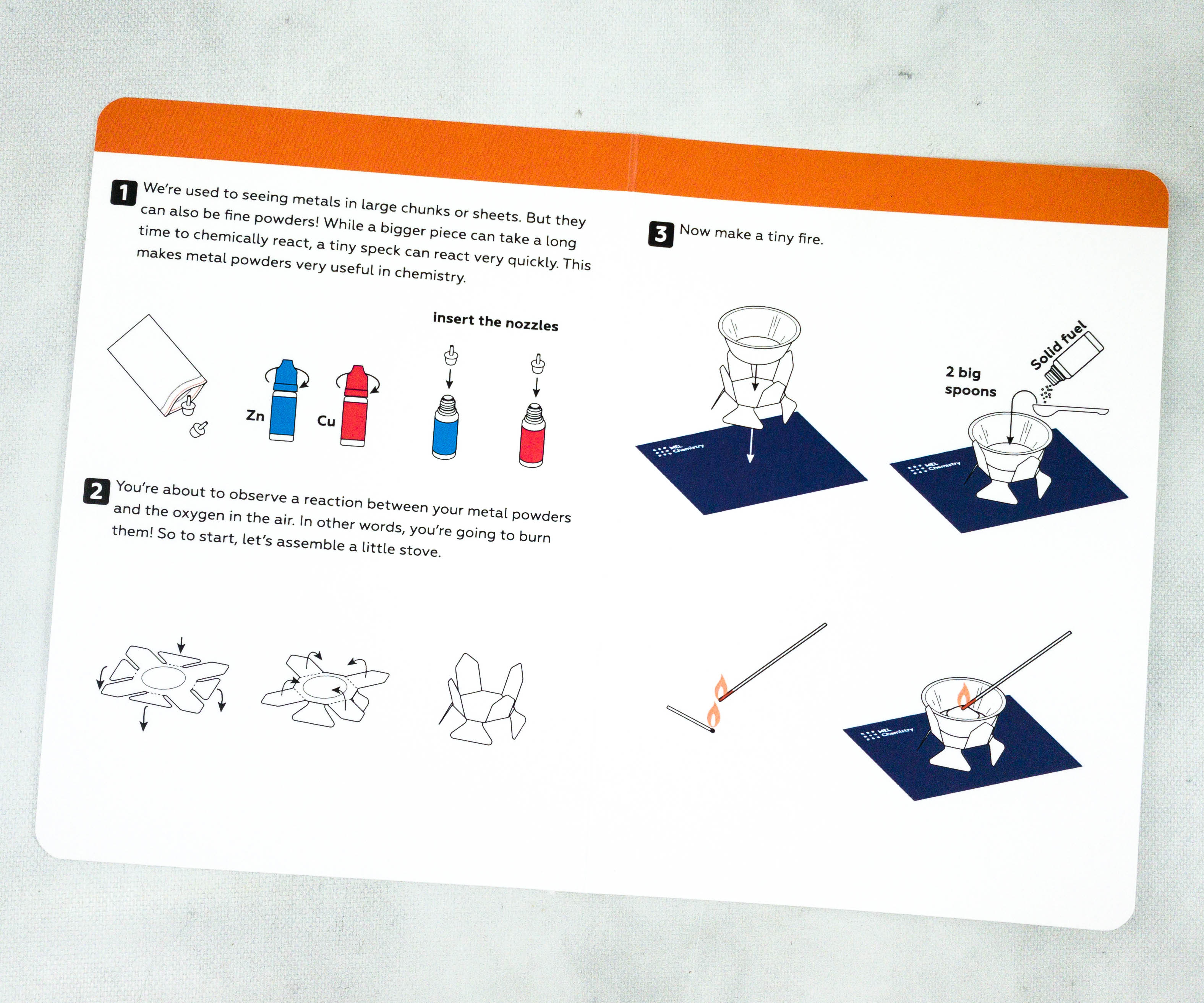
Inside, you’ll find the complete and illustrated instructions for the experiment!

Copper powder, hexamethylenetetramine (solid fuel), and zinc powder are the reagents for this experiment.

The activity comes with an instruction and information sheet.

This sheet provides a few reminders, a list of reagents, instructions for adults, and proper disposal.

It’s important to set up the materials properly for a safe and fun experiment experience!

For the first step, my daughter placed Hexamethylenetetramine into the aluminum mold using a measuring spoon.

The wooden stick is used instead of the regular sized match to have a safe distance in dealing with fire.

Here, my daughter used the stick to start a flame using the solid fuel.

It’s also important to have water nearby as we perform the experiment.

Once the source for the flame is set, it’s now time to put the metal powders and observe how each reagent reacts when placed on it.

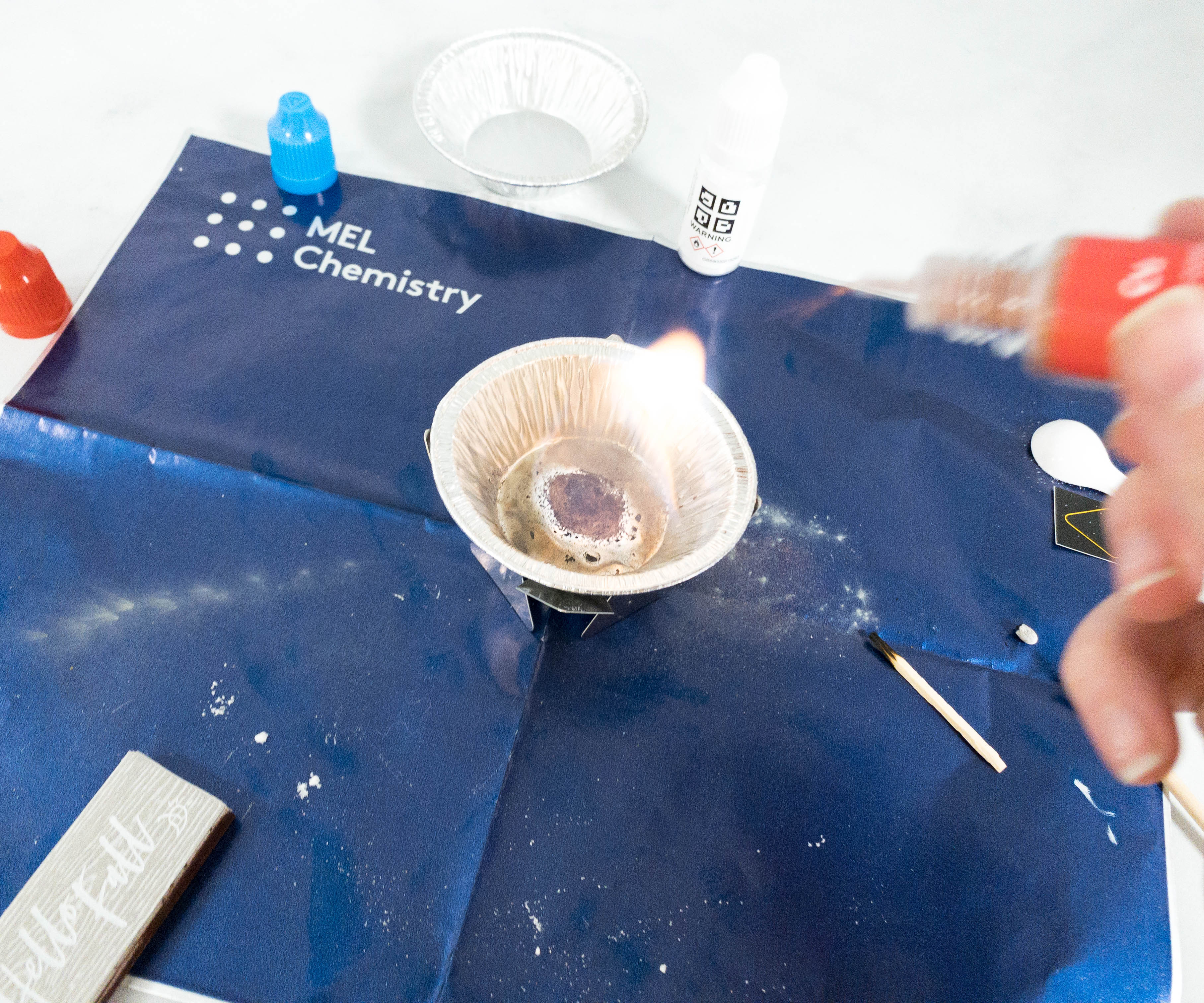
Next, my daughter put zinc powder into the source of fire.

The color of the flame turned into blue when the zinc powder started burning.

It went back to its original color when all of the powder was consumed by the flame.

She had to wait until all the solid fuel burns out completely. It’s not advisable to add more while the source is still burning.

Once the flame is stable, you can proceed with the experiment.
This time, my daughter added copper powder to reveal a different chemical reaction.
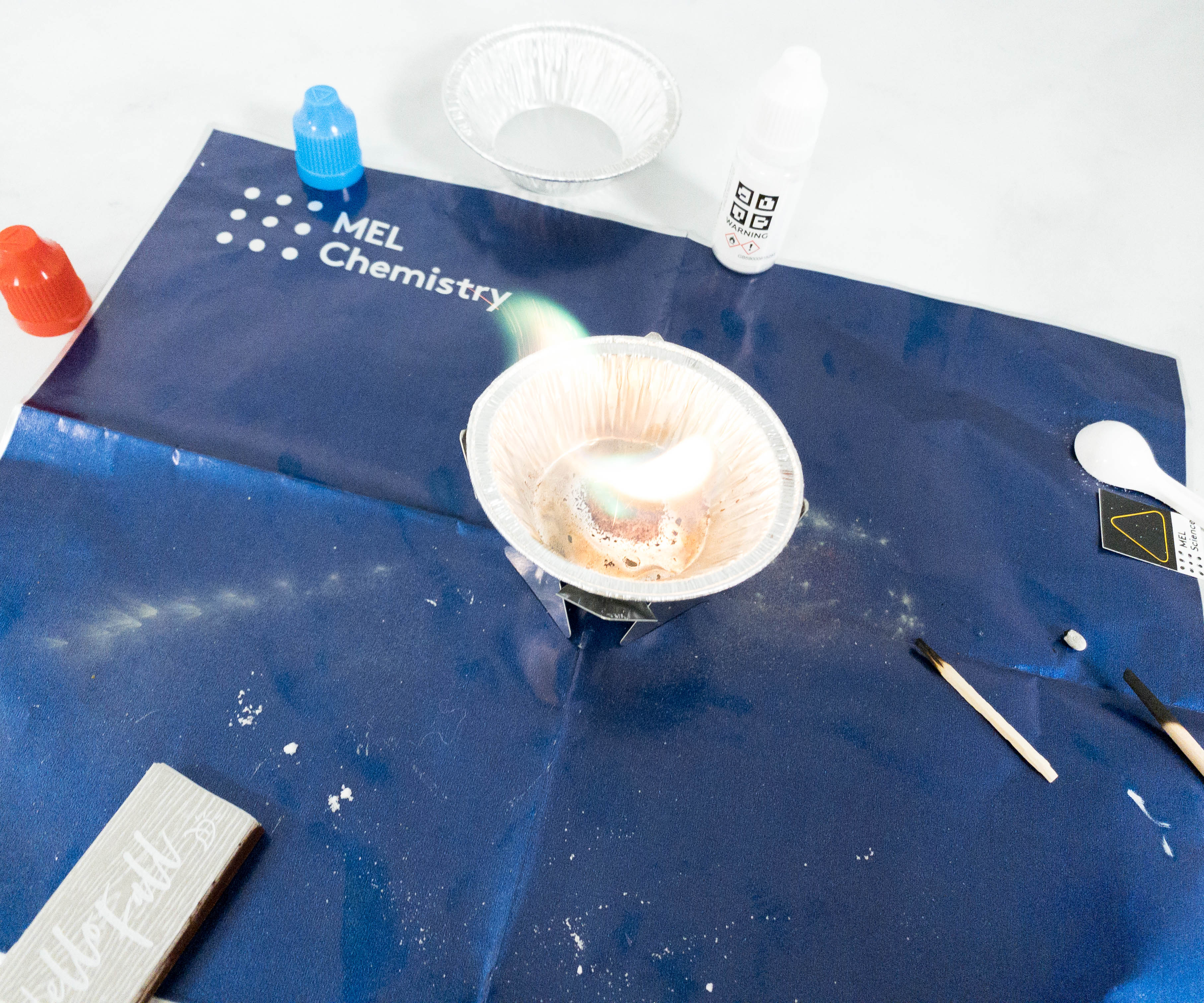
This reagent reacted immediately after it started burning.

This time, the fire produced a green color. As expected, it reacted differently compared to the zinc powder!
My daughter had a great time with this experiment from MEL Lite by MEL Science (and I promise – she used ALL the fuel because she couldn’t get enough of this one!)! The reagents produced different colors when burned because they didn’t have the same atoms or ions that were emitted during the process. Everything went smoothly because the instructions are clear and well-detailed. Since this experiment deals with fire, there were enough warnings and reminders mentioned on the information card and sheets that help give kids a safe experiment experience. Overall, this is science subscription box is always well-put-together so I couldn’t ask for more! It’s ideal for kids who enjoy science and experiments, and also for parents who want to nurture their kids’ love for science!
What do you think of the MEL Chemistry Lite by MEL Science box?
Visit MEL Lite by MEL Science to subscribe or find out more!




Comments