
MEL Chemistry is a monthly AR‑powered STEM subscription box from MEL Science intended for kids ages 9-14. You can subscribe to the box for $34.90 each month. Each monthly kit contains 2-3 science experiments with enough materials to try it twice, plus step-by-step instructions. All the experiments are designed for home use.
DEAL: Get 25% off your first month! Use coupon code HELLO25.

This is the review of the MEL Chemistry – Chemistry of Plants box.

The box welcomes us to science in different languages!
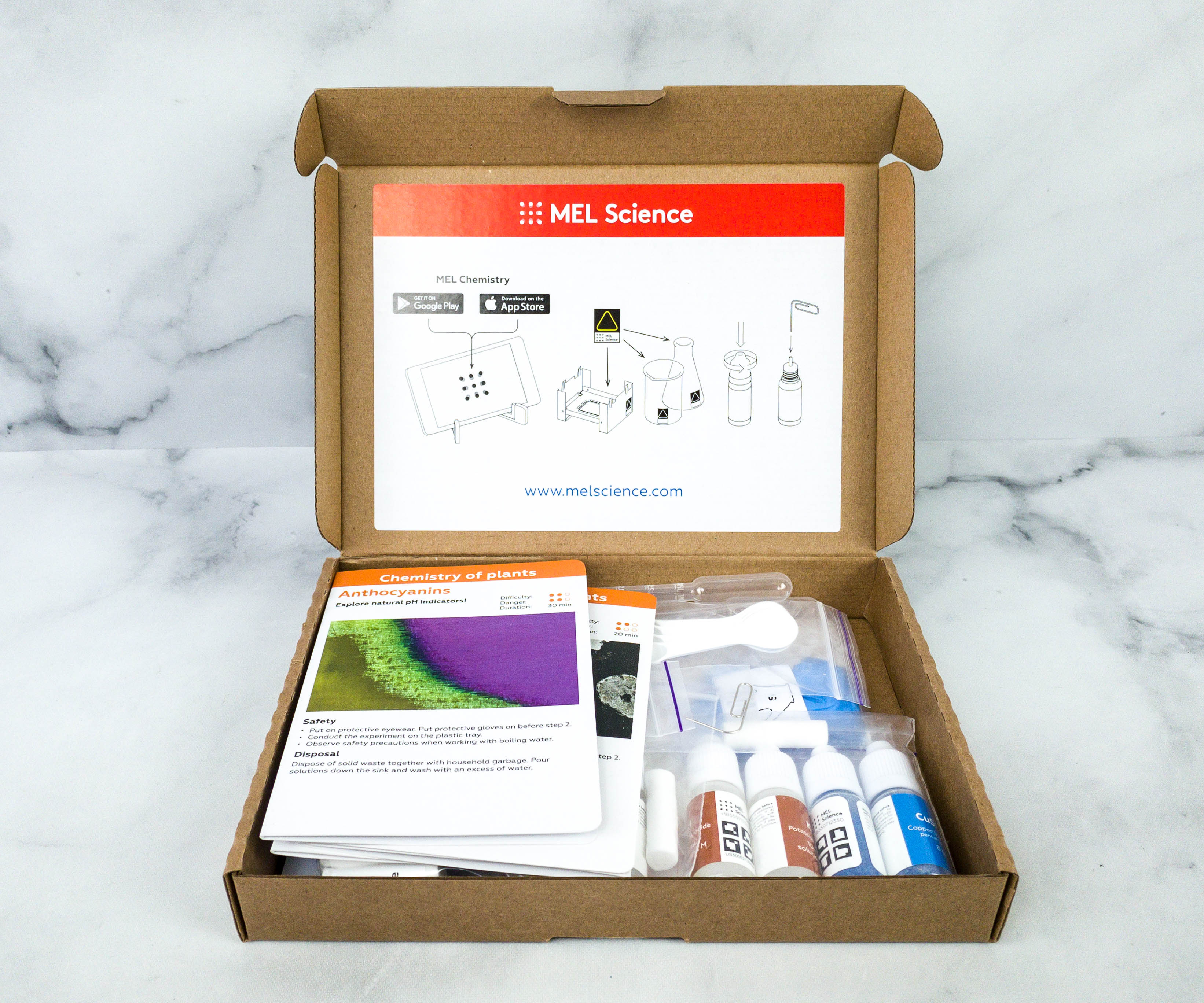
We’re so excited to finally open this box!

To help manage our experiment, they encourage us to install the MEL Chemistry app on a smartphone. It also has an instruction on how to put a hole in the bottle’s nozzle and where the kids can place the MEL stickers.
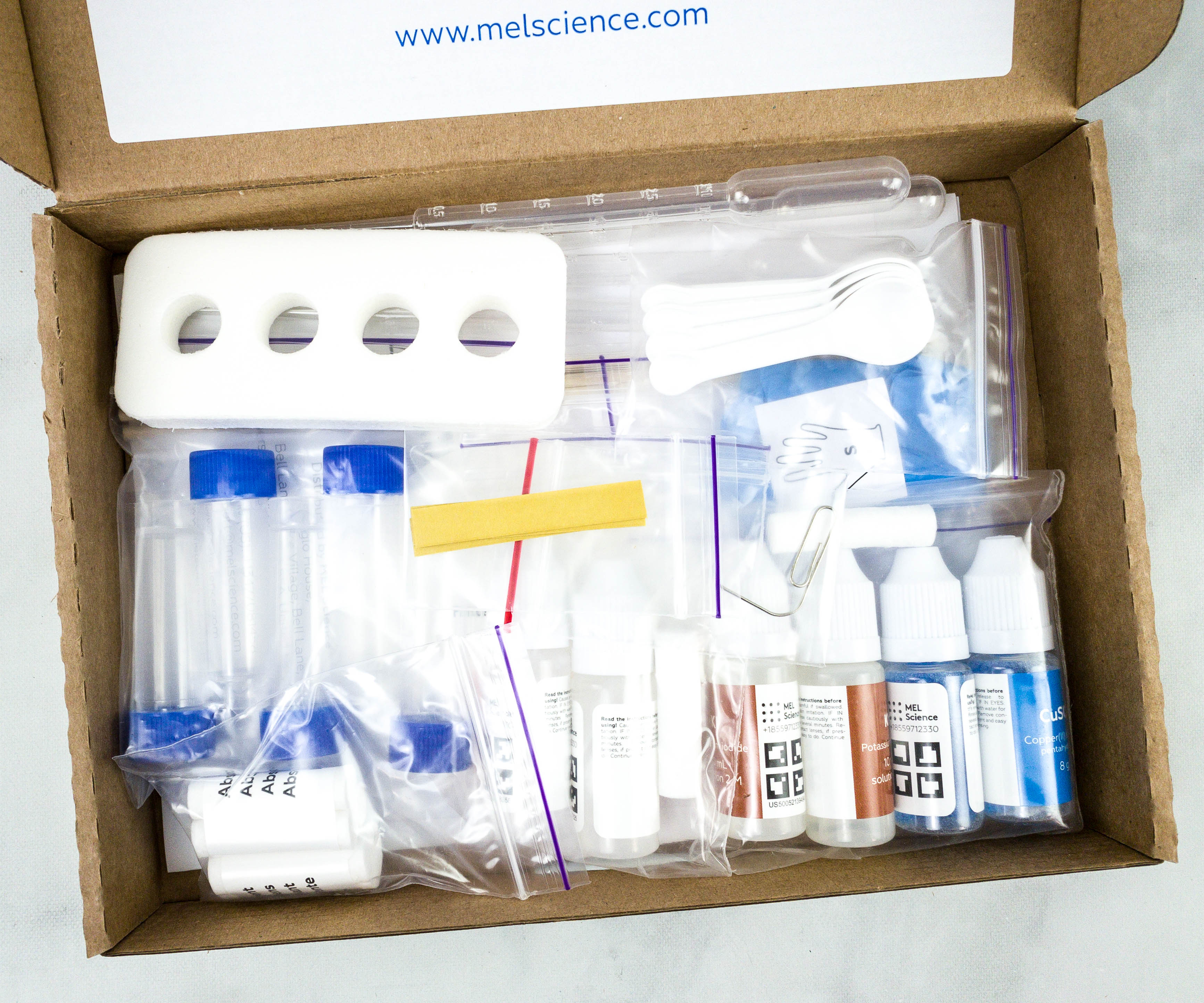
All the materials were well packed.

The activity comes with an instruction and information sheet.

This sheet provided us with a few reminders, a list of reagents, and instructions for adults.

The paper listed the instructions for each activity. It also reminds us how to properly dispose of the waste after every experiment.

Safety is always a priority when doing experiments!

Everything in the box!

Some of the materials were packed in individual plastic pouches.

Herer are our reagents for this month’s experiments.

Each activity came with its own booklet. There’s a total of 3 experiments with this box!

Activity #1: Anthocyanins. Our first activity is about Anthocyanins and exploring natural pH indicators.

Chemists use pH as an acidity unit. We used pH indicator strips in this part of the activity.
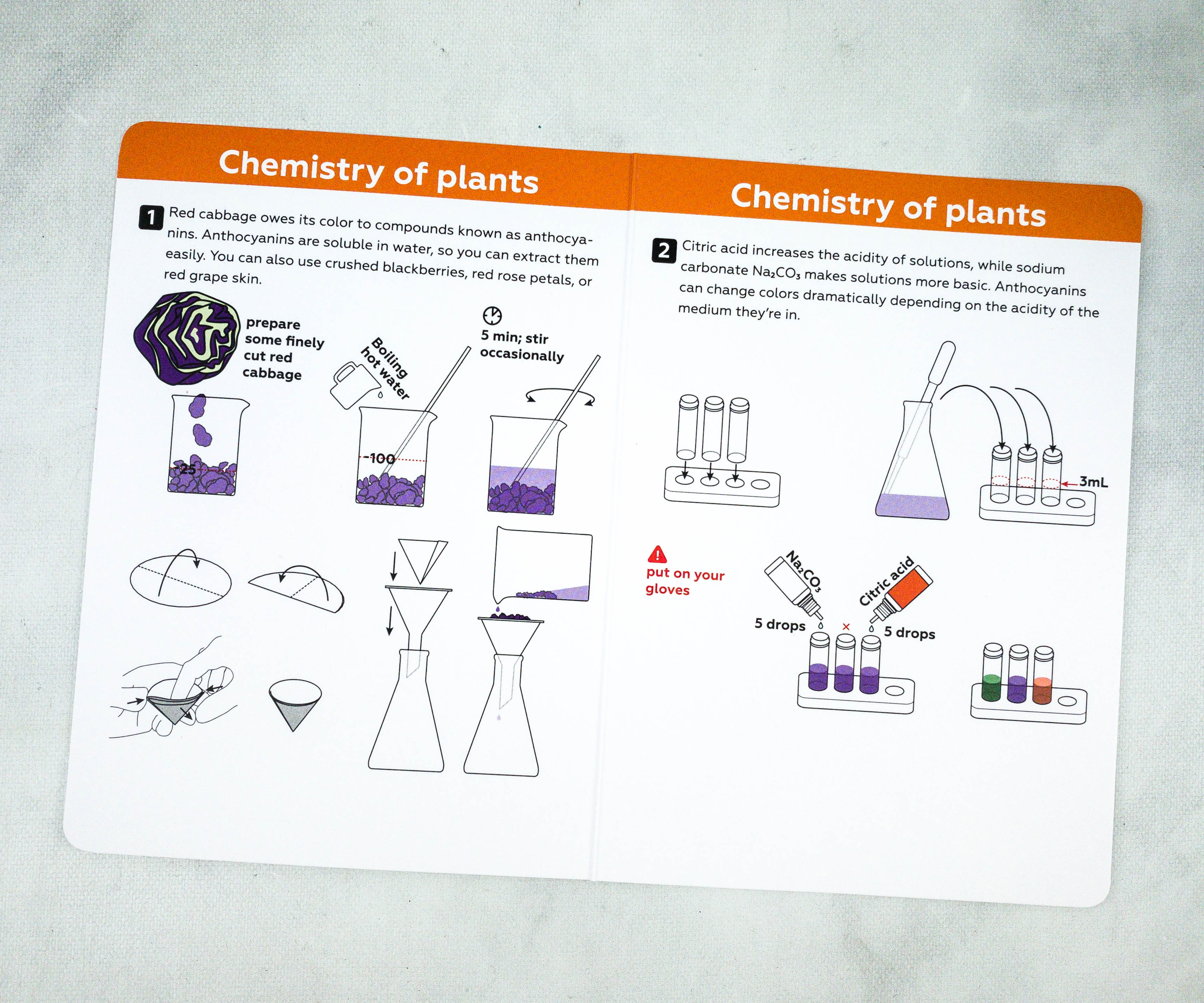
This card contains instructions and illustrations for our first experiment.

Here’s the story about “The Violet Accident” wherein scientist Robert Boyle and his assistant accidentally discovered how flowers and plants change colors when exposed to acids and alkalis.

Anthocyanins is a whole family of plant pigments that are also found in fruits, vegetables, and some leaves. It helps protect the plants in cold or hot weather.
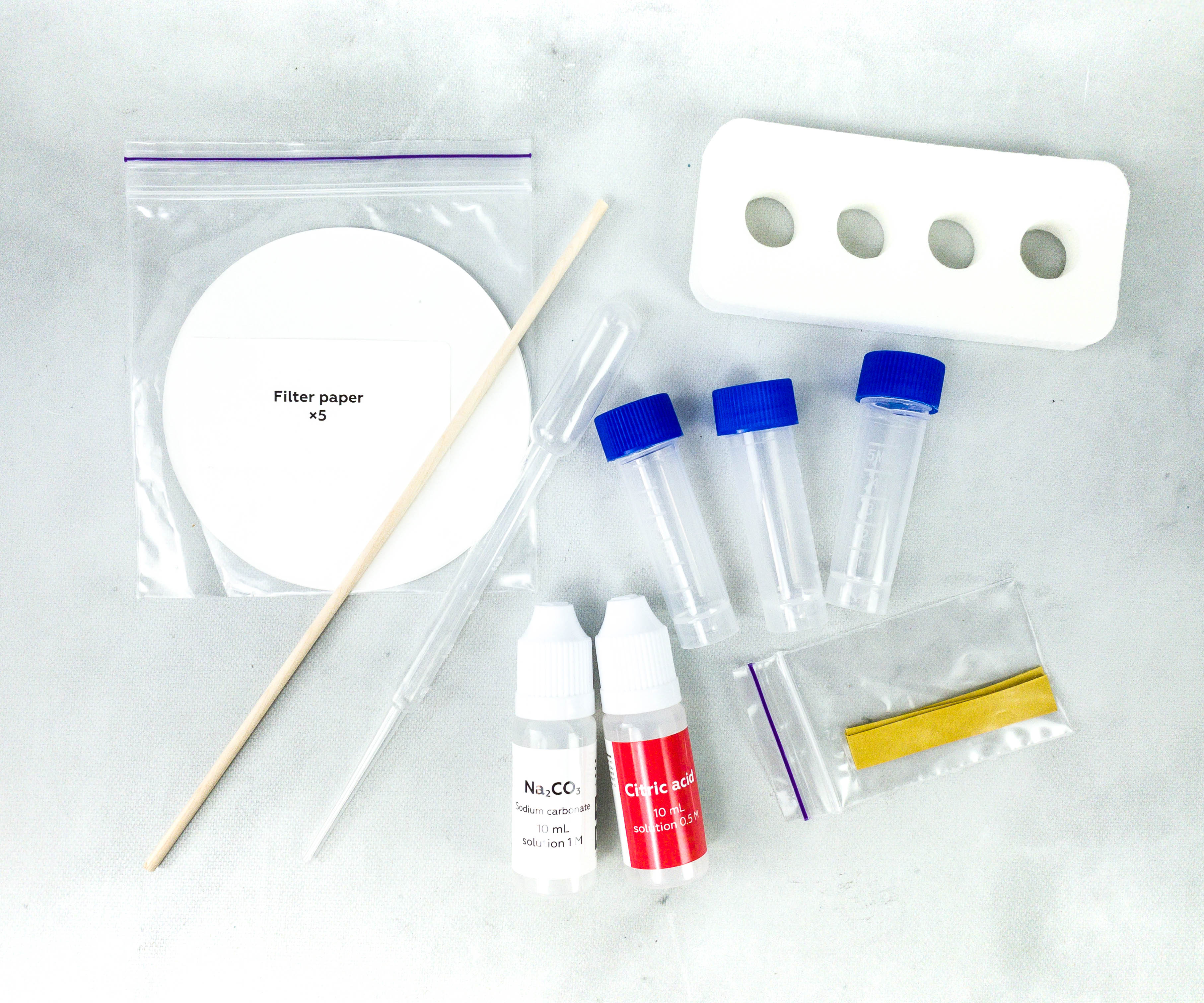
The box also includes filter paper, wooden stick, pipette, indicator strips, and vials.
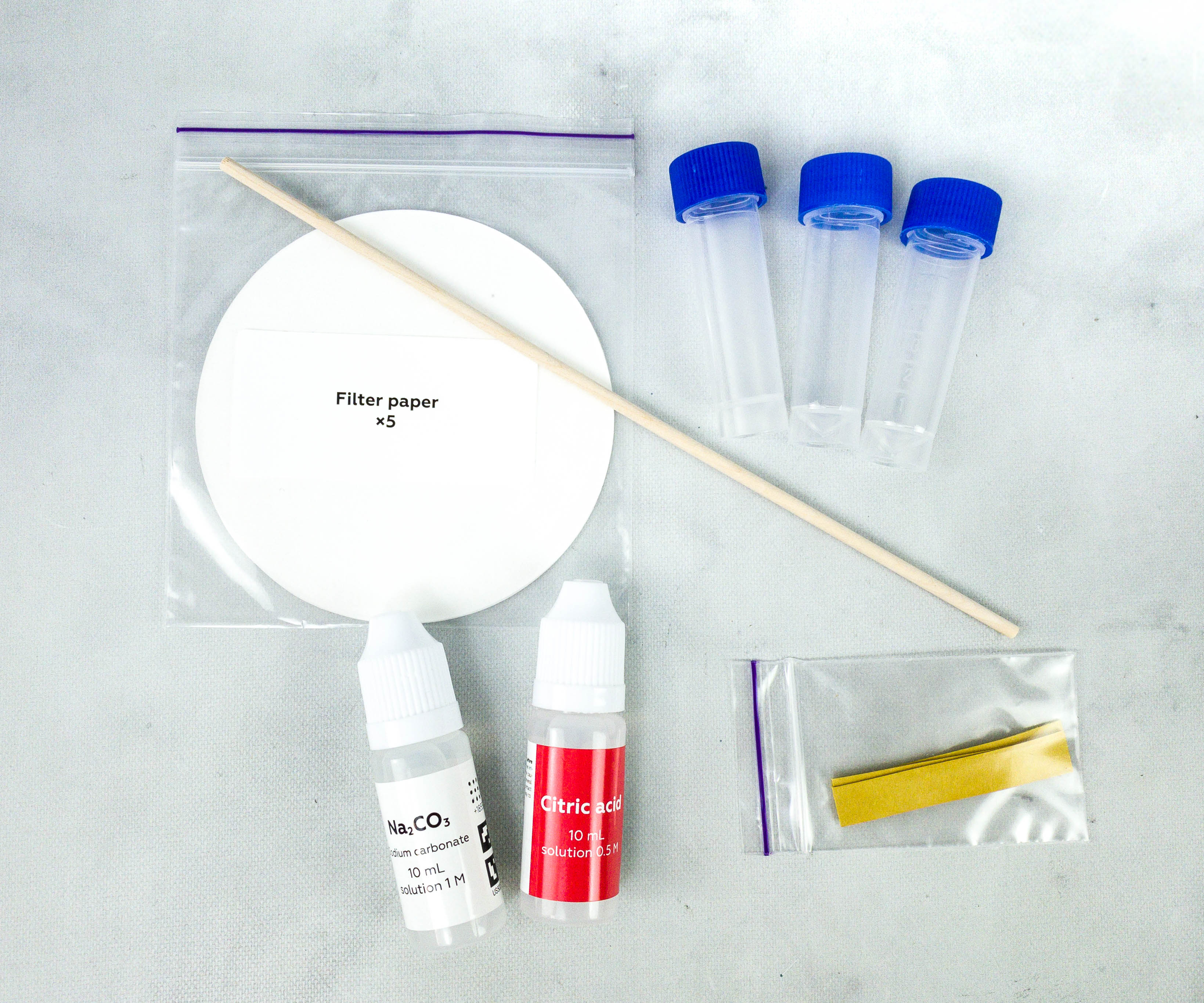
The reagents, sodium carbonate and citric acid, are also placed in individual bottles.
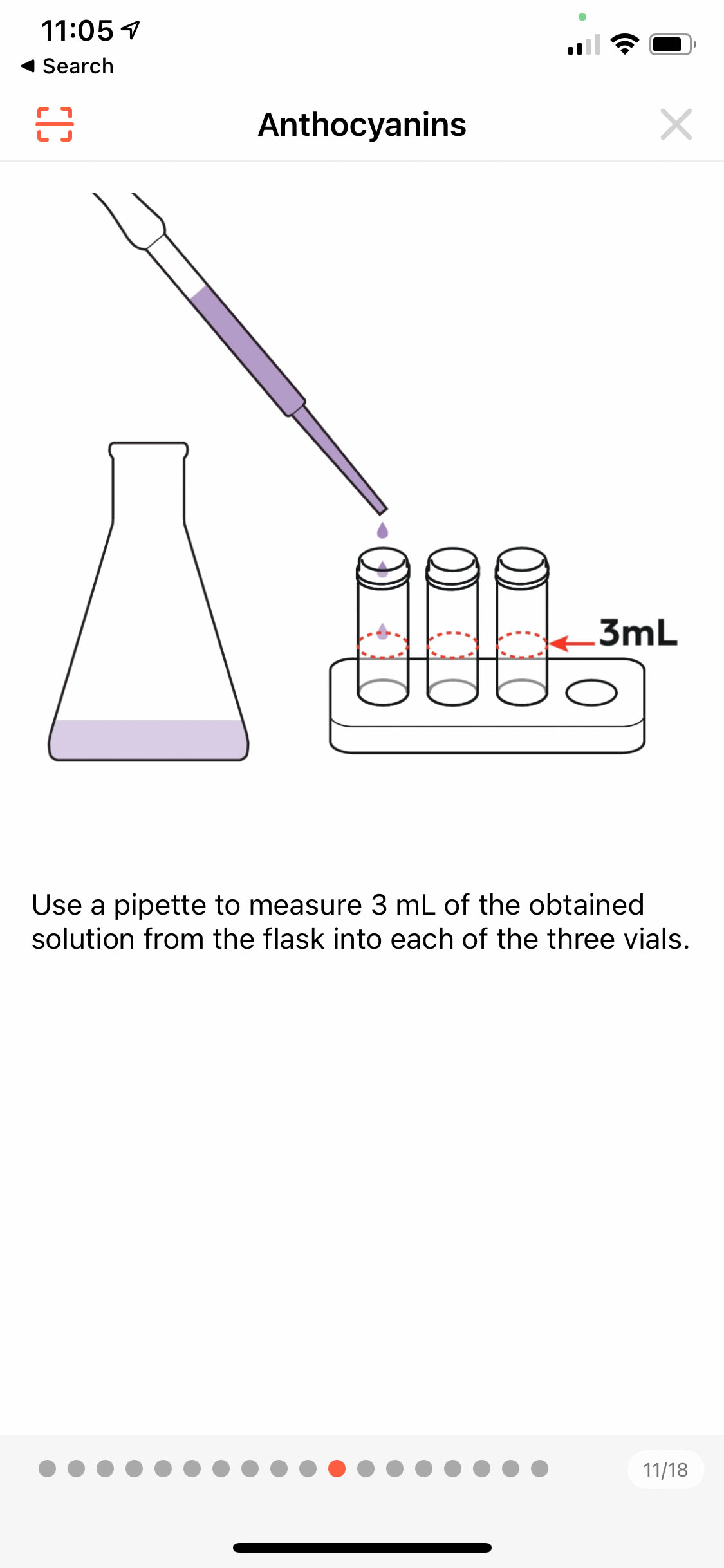
The app has an illustration of how to transfer a solution from the flask into each of the 3 vials using a pipette.

As instructed, the kids have prepared a 3ml cabbage solution on each vial.
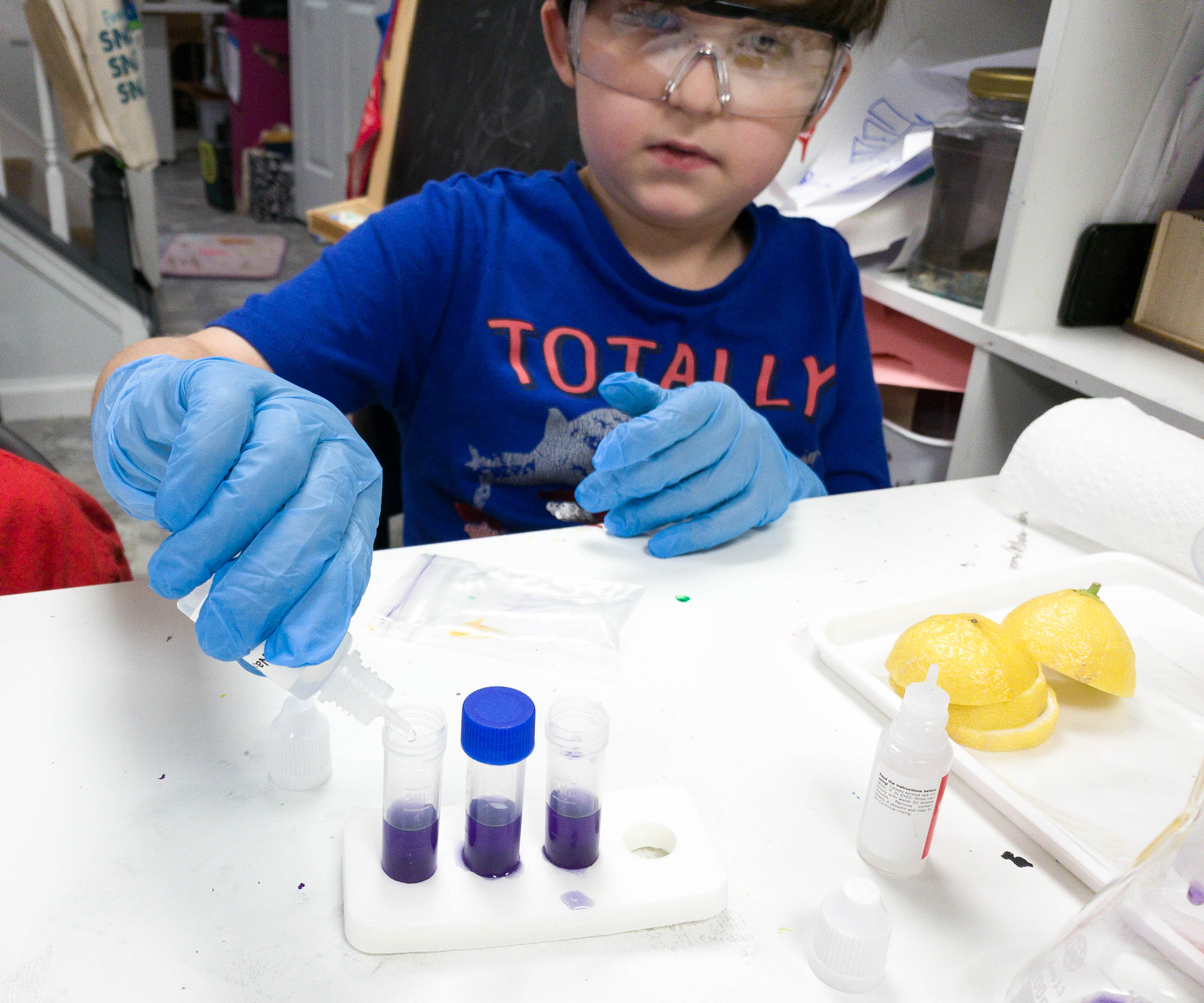
Sodium carbonate is then dropped into the solution. Let’s see how our solution reacts to it!

It’s starting to change its color! The solution is slowly reacting to our reagent.

There are now green, blue, and purple colors on the vials!

This pH indicator strips also changed its color!

It’s still reacting even after a few seconds.

The pH strips reacted differently depending on the acidity of each solution.

My kids compared the colors of each strip and referred to the booklet to check the results.

Activity #2: Plants vs. Iodine. With this experiment, the kids are about to discover which food item has ascorbic acid in it!

At the back, the importance of vitamin C on our bodies is discussed and what happens if we lack it.
Detailed instruction with illustration is also provided for every step of the process.

After the instructions, there’s also a brief explanation of how our experiment works!

The Great Oxygenation Event is also discussed in the app. It’s an event that happened some 2.5 billion years ago wherein almost every living thing on Earth was killed. This article explains that oxygen is a strong oxidant and can take electrons from other compounds, which can make or take life from a living organism.
There are lots of other fruits and vegetables that kids can also try with the experiment, like tangerines, grapes, and apples!

These are the materials for the plants vs. iodine experiment! The reagents are potassium iodide and copper (II) sulfate pentahydrate.

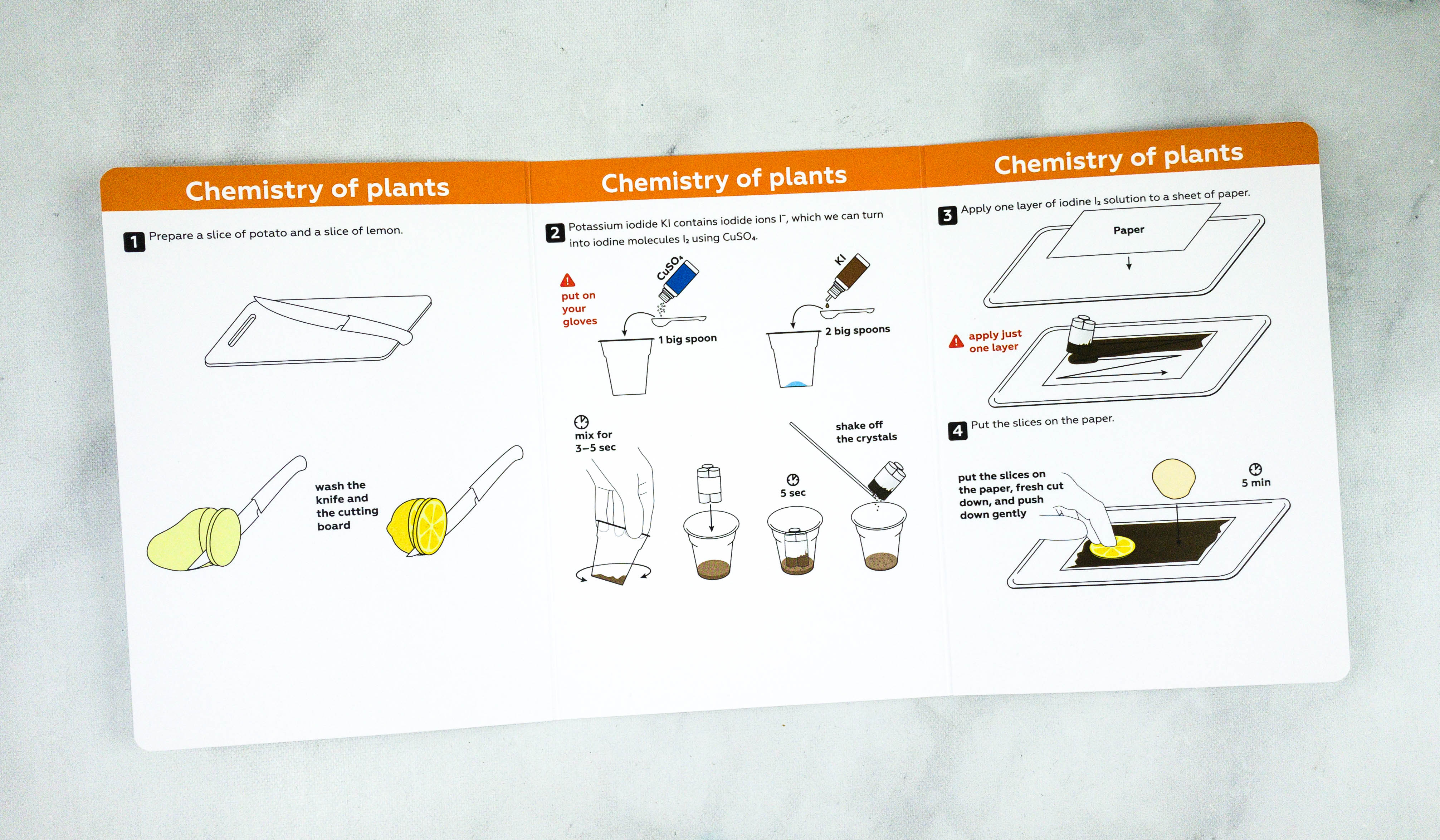
My kids mixed potassium iodide and copper sulfate pentahydrate.

After mixing, they applied the solution on a piece of paper.

They need to cover the whole sheet with the solution.

The next step is to place the potato and lemon slices on top of the sheet.

Push the slices down gently into the piece of paper to get more visible results!

It’s now time to check the results! Let’s see how the mixture reacted with the potato and lemon!

The more ascorbic acid the fruit or vegetable has, the more iodine it can turn into iodide ions, and the less brown iodide will be left on the paper.

With the results, it’s clear that the lemon has more vitamin C in it!

Activity #3: Is This Food Starchy? The last experiment is to detect starch in plants! My kids also used slices of potatoes and lemons for this experiment.

In this experiment, the kids determined which of the two contains starch by adding 5 drops of iodine to each cup.
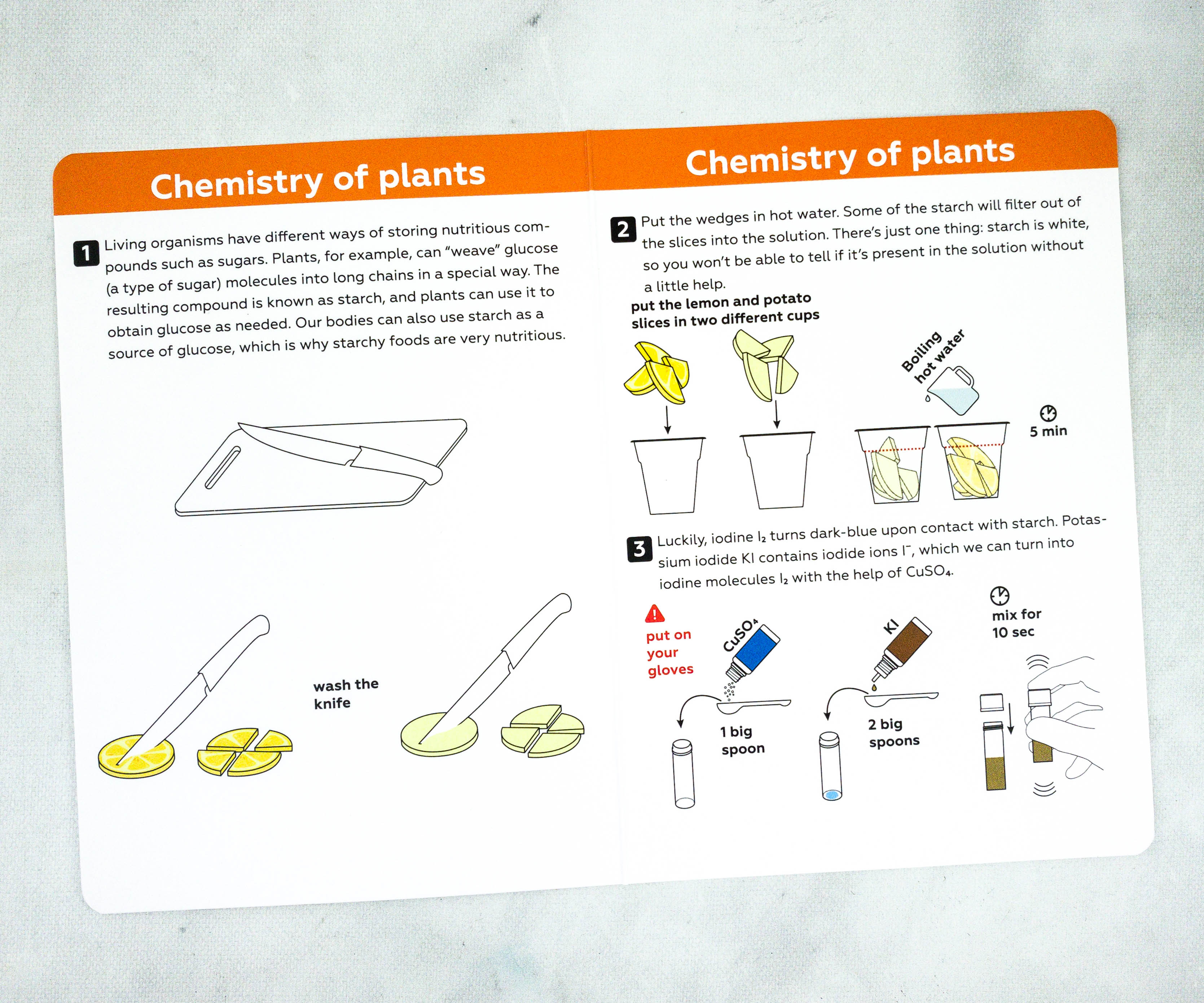
Instructions with illustrations are available for this experiment as well.
My kids used the timer to perform the activity with starch. It’s important to follow instructions to achieve the correct result!
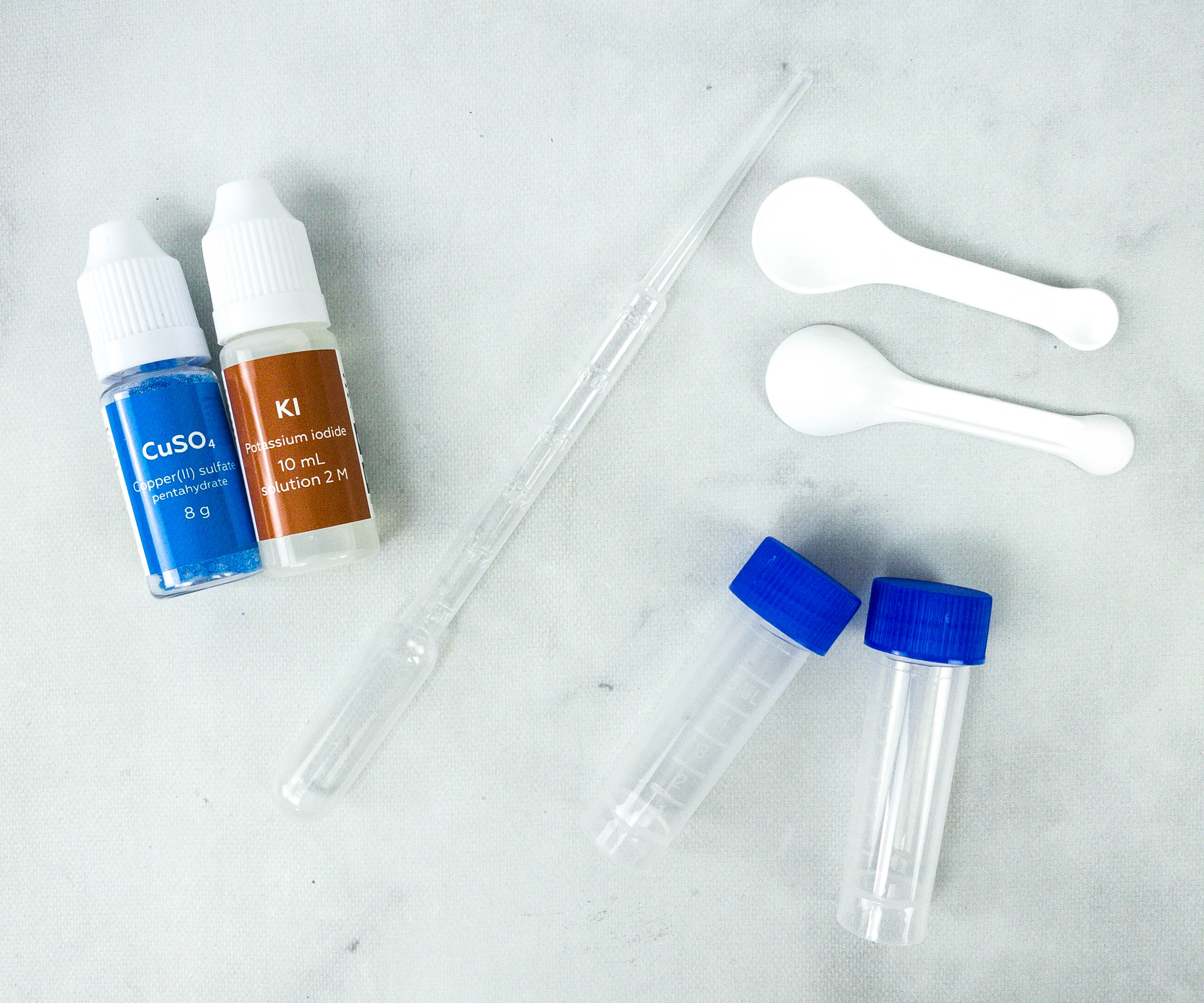
These are the materials and reagents my kids used in the last experiment.

The first step is to pour the boiling water on the cups with sliced potatoes and lemons.

Using the filter paper, they got a pure extract from the cabbage solution.

To make the iodine solution, they put 1 big spoon of copper sulfate pentahydrate into a vial.

Then, my son also added 2 big spoons of potassium iodide. The solution is then mixed for about 10 seconds.

They can now extract the solution using a pipette!

Here’s the next step!

It’s now time to put 5 drops each on the potato and lemon cups.

The potato is reacting to the iodine solution. It’s turning blue while the lemon is showing no results.

The water in the potato cup changed into a dark blue color. This experiment shows that there’s starch on the potato!
This month’s MEL Chemistry is a triple treat for my kids! The 3 experiments provided them with new knowledge about the chemistry of plants. It’s nice that most of the materials are included in the box, and the other materials needed from home, like the potatoes and lemons are easily available. As always, they provided easy to follow instructions! My kids really enjoyed and were surprised by the results! This is a great subscription that allows the kids to learn about chemistry and do experiments at home!
What do you think about the MEL Chemistry subscription ?
Visit MEL Chemistry by MEL Science to subscribe or find out more!




Comments